Exploring the gel phase of cationic glycylalanylglycine in ethanol/water. II. Spectroscopic, kinetic and thermodynamic studies
Abstract
Hypothesis
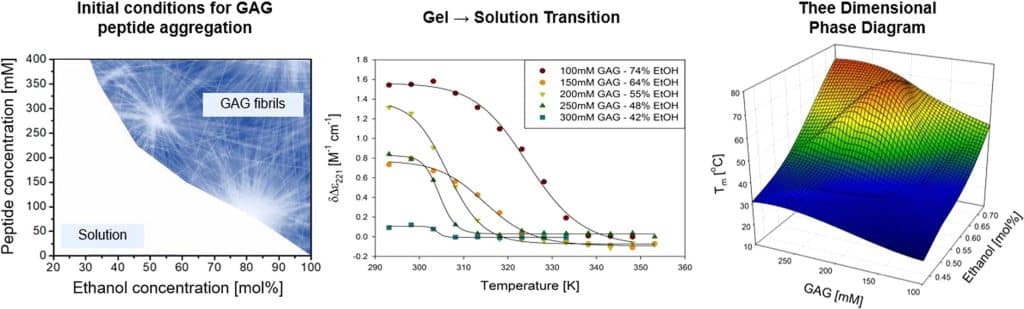
Recently, we reported a three-dimensional phase diagram for the gelation of cationic tripeptide glycylalanylglycine (GAG) in water–ethanol mixtures. We showed that the gel strength reaches an optimum for a peptide concentration of 200 mM and ethanol/water mixtures with ca. 55–60 mol% ethanol. An increase of the ethanol fraction causes a substantial upshift of the gel’s softening temperature which is indicative of a reduced peptide solubility. We expect the formation of long crystalline fibrils which form the sample spanning network of the gel phase to precede the gelation process and that the fibril microstructure depends on the rate and concentration of peptide.
Experiments
We used UV circular dichroism (UVCD) spectroscopy to probe the kinetics of GAG fibril formation as a function of peptide concentration and ethanol fraction. We provide experimental evidence for the notion that the utilized CD signal reflects the three-dimensional assembly of peptides rather than a two-dimensional sheet structure. UVCD was also used to probe the melting of GAG fibrils with increasing temperature. FTIR and vibrational circular dichroism (VCD) spectroscopy were employed to characterize the structure of sheets with which the observed fibrils were formed.
Findings
Fibrilization and gelation kinetics occur on a very similar time scale for very short gelation times (<7 min) observed at high peptide concentrations and/or ethanol fractions. Otherwise, gelation proceeds significantly slower than fibrilization. The trends in the UVCD spectral response parallel the trends in the storage modulus as a function of peptide concentration and ethanol fraction. IR and VCD profiles of amide I′ reveal that fibril structure and the respective chirality are both affected by peptide concentration and solvent composition. At high ethanol fractions, the VCD changes its sign suggesting a conversion from phase II to phase I. Generally, the latter is obtained only at temperatures below 15 °C. Altogether, our results reveal how GAG fibrilization and gelation are interrelated and how the gel properties can be tuned by changing the composition of the ternary GAG/water/ethanol mixture.
Publication Metadata
Authors
David M. DiGuiseppi; Lavenia Thursch; Nicolas J. Alvarez; Reinhard Schweitzer-Stenner
